Octet software is easy to learn and has an intuitive interface with powerful data analysis features. The latest release, Version 11, is our most comprehensive solution yet and is available for use on all Octet systems. For more information or upgrade options, please fill out our software download request form or contact your local sales representative.
New features include:
- Analyze multiple plates and experiments together for all analyses in Octet Data Analysis HT software, reducing analysis time from hours to minutes
- Customize PDF data reports using text, graphs, data tables and images
- More flexible and advanced data processing and correction options to choose from
- New graphical display options to thicken sensor traces and customize axis scale and title options
- Enhanced features for confident 21 CFR Part 11 compliance
- Automated analysis enabled for quantitation, kinetic and epitope binning data
- Compatible with 64-bit versions of Windows® 10
Design and Analyze an Assay in Minutes
A new assay can be set up in just a few minutes. To make assay set up even faster, experimental method templates are available. Analysis settings can be saved and applied to a new assay to significantly speed up routine analysis.
Powerful Analysis that Gets you Answers Quickly
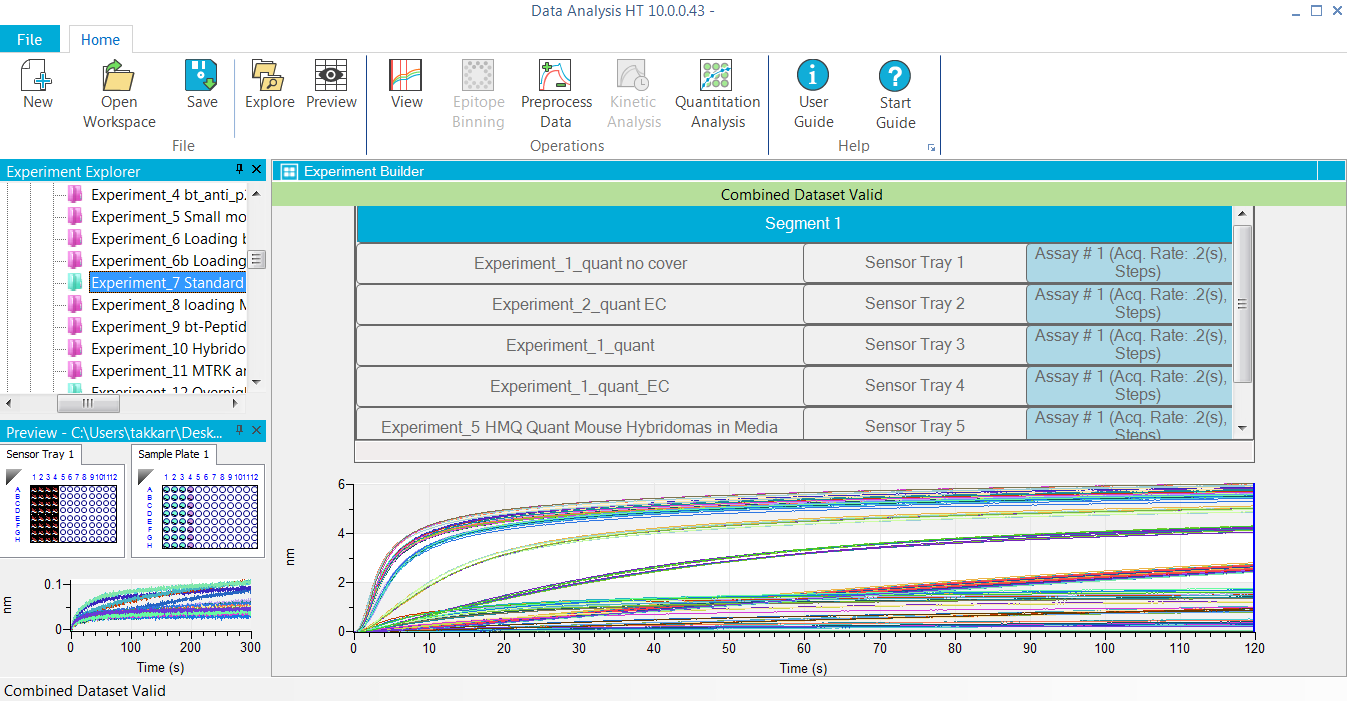
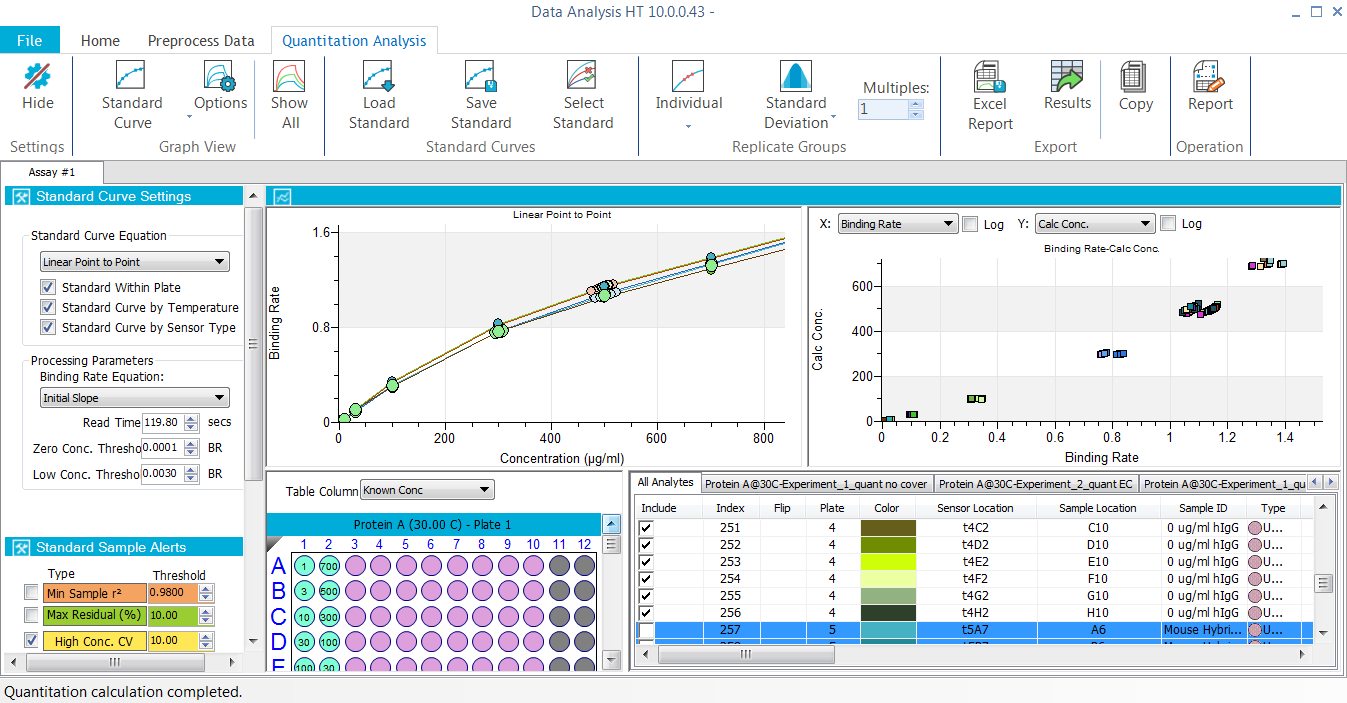
Quantitation Assays
- Applications: Quantitation of total IgG, Fab and custom proteins, contaminant testing such as host cell protein (HCP) and residual protein A, pharmacokinetic (PK) assays, antibody isotyping in complex samples, glycosylation profiling, vaccine titer determination etc.
- Combine and analyze multiple experiments and plates together
- Multiple and flexible reference subtraction
- Linear, 4 and 5-parameter logistic (PL) unweighted and weighted fittings
- View and analyze any step(s) in a multi-step ELISA-type assay
- Automated sample alert tools to evaluate data quality for precision, accuracy and dilution linearity, enabling you to interpret results with confidence
- Import previously made standard curves for routine assays
- Customize and export final reports in Microsoft® Excel® and PDF formats
Kinetic Assays
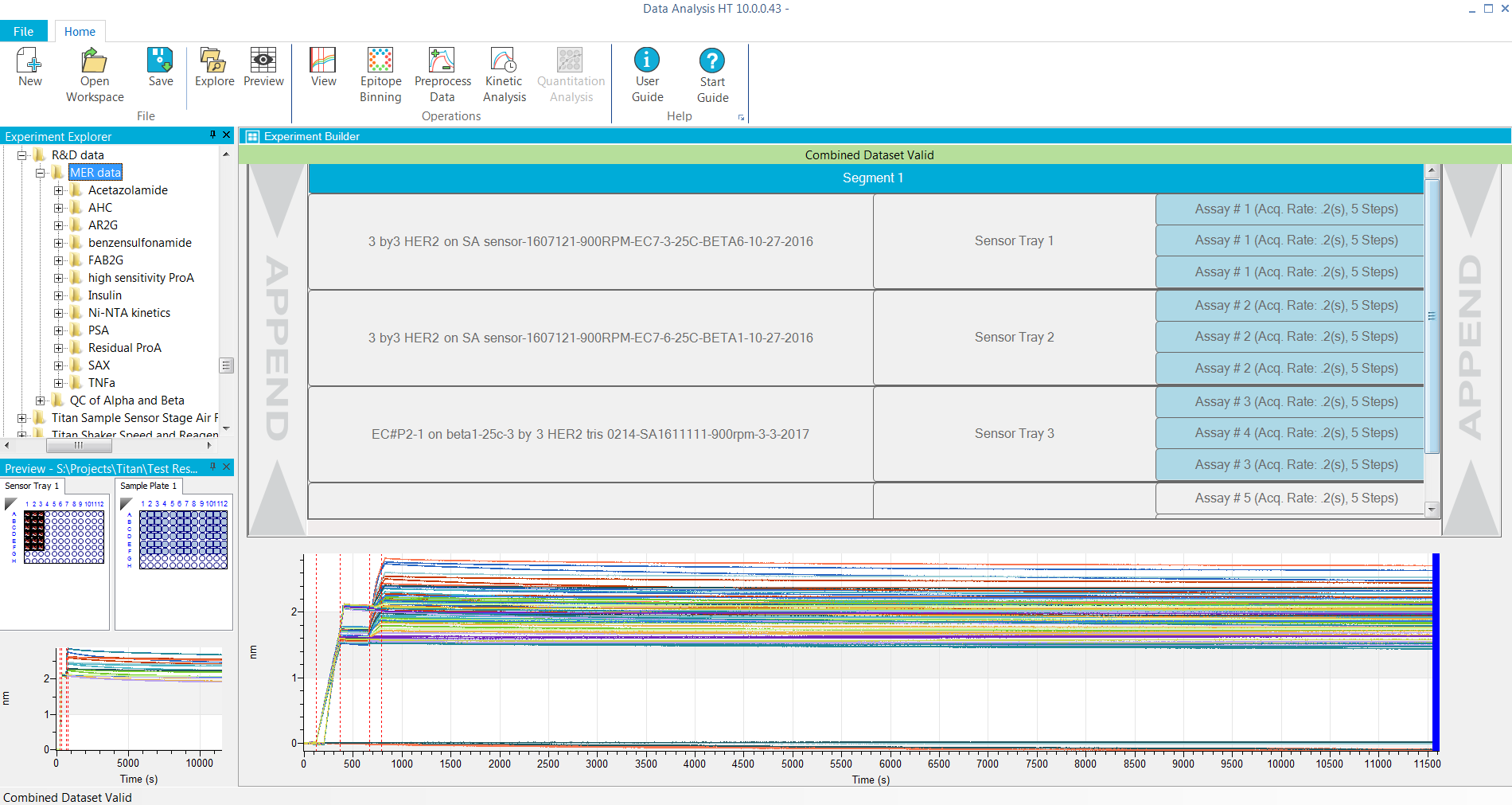
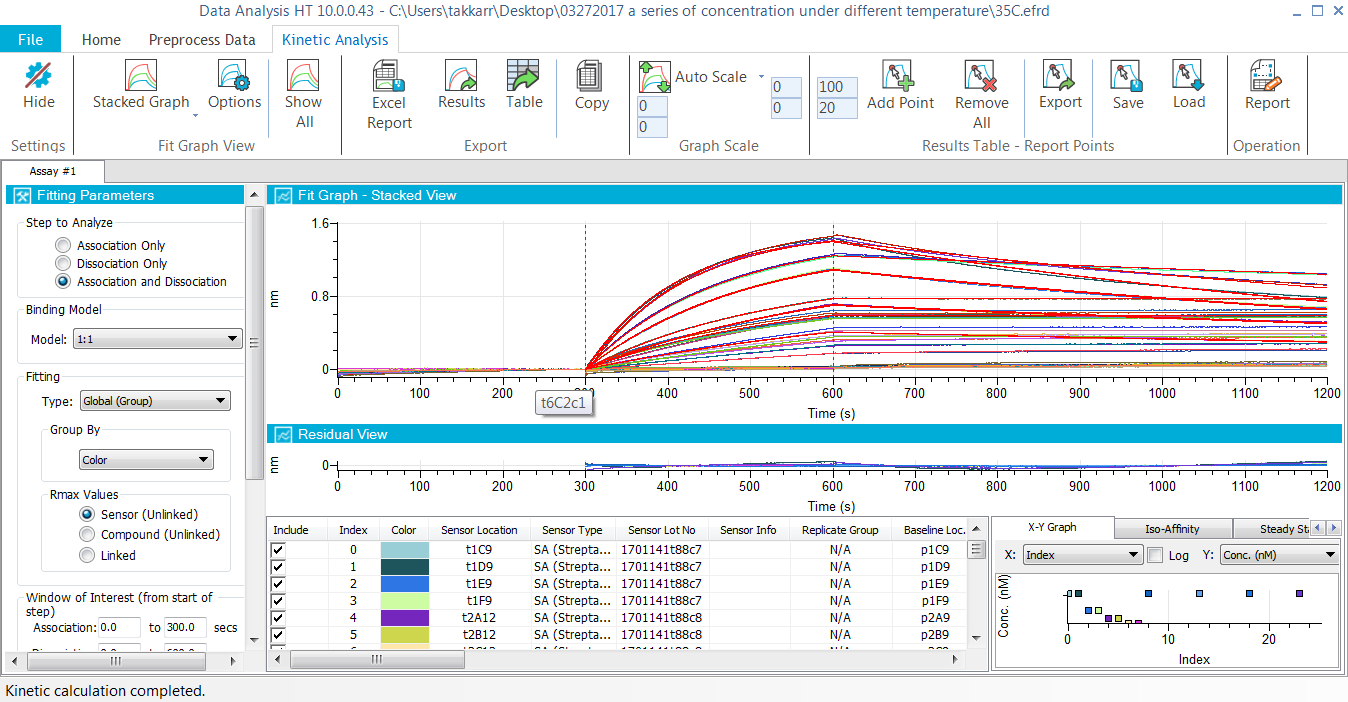
- Applications: Ligand/target binding, kinetic screening (off-rate or affinity) in supernatants and purified samples, kinetic analysis of protein-protein, protein-small molecules, antibody-antigen, antibody-VLP, antibody-Fc gamma receptors, antibody-neonatal receptor (FcRn) interactions. Lot release and stability testing of drug molecules can also be analyzed using Octet software.
- Combine and analyze multiple experiments and plates together
- Flexible and additional reference sample and biosensor subtraction
- Advanced data processing options remove bulk shifts or assay artifacts
- Faster and improved 1:1, 1:2, 2:1 and mass transport curve fitting models
- Local (partial or full) or Global data fitting options
- Steady-state analysis
- Add report point(s) to any step in the sensor trace for screening purposes
- Customize and export final reports in Excel and PDF
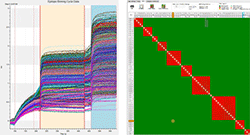
Epitope Binning Assays - Advanced Analysis with HT Software
- Append and overlay data from multiple experiments, plates or biosensor trays to create a combined dataset for advanced analyses
- Software automatically finds the binning cycle pattern and creates a 2-D traffic-light matrix for you
- Cluster antibodies in similar bins or groups using custom (Euclidean, Pearson, Maximum) algorithms
- Additional matrix analyses like normalizing to controls or self-binding and highlighting unidirectional binning pairs can be performed
- Simple data quality assessment can be accomplished by monitoring loading levels and flagging antibodies with high off-rates
- Customize and export final reports in Excel and PDF
Customized Report Generation
- Create customized reports for experiments, combining various data elements such as graphs, text, data tables, company logo, images and experimental details
- Reports are ready to be uploaded to an electronic notebook or stored in the database
- Report templates can be saved and re-loaded to make similar reports for additional datasets
21 CFR Part 11 Tools for Confident Compliance
Octet CFR software and our GxP Server enable data acquisition and data analysis in laboratories working under GMP, GLP, and 21 CFR Part 11 regulations. It also provides all necessary technical administrative features for compliance with FDA regulations.
- Controlled access with multiple user levels and a set of permission structure — administrator, developer, supervisor, lab user
- Secure primary data integrity — digitally signed acquired data that is invalidated if tampered
- Electronic signatures — enable data to be locked after analysis is complete
- Enhanced Audit Trail — all actions are recorded and time-stamped with details of old vs. new values
- Software Validation Package — verifies calculations in Octet software with third-party software to trim validation time to just 3 days
For more information on each software version, please refer to the or .